Water Liquid-Vapor Pressure vs Density simulated by GCMC
- FarawayK
-
 Topic Author
Topic Author
- Offline
- New Member
-

Less
More
- Posts: 6
- Thank you received: 1
5 years 6 months ago #539
by FarawayK
Hi Ryan,
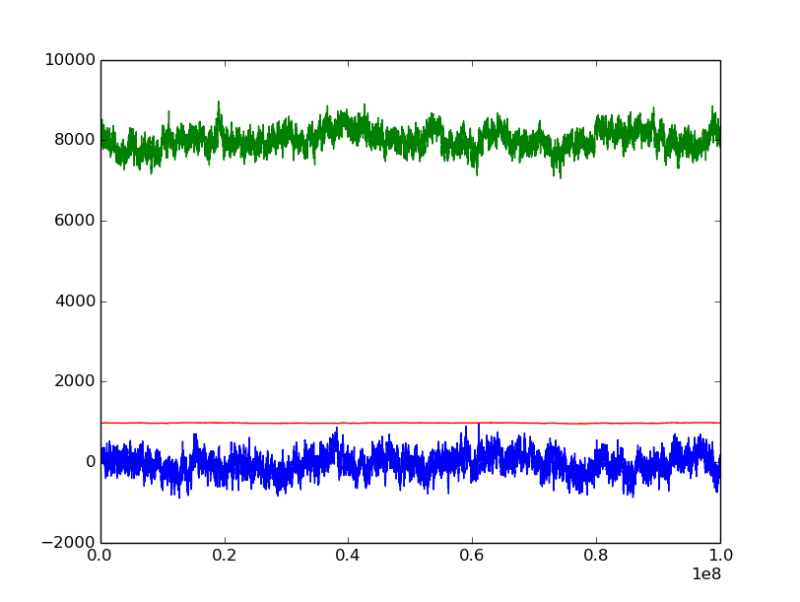
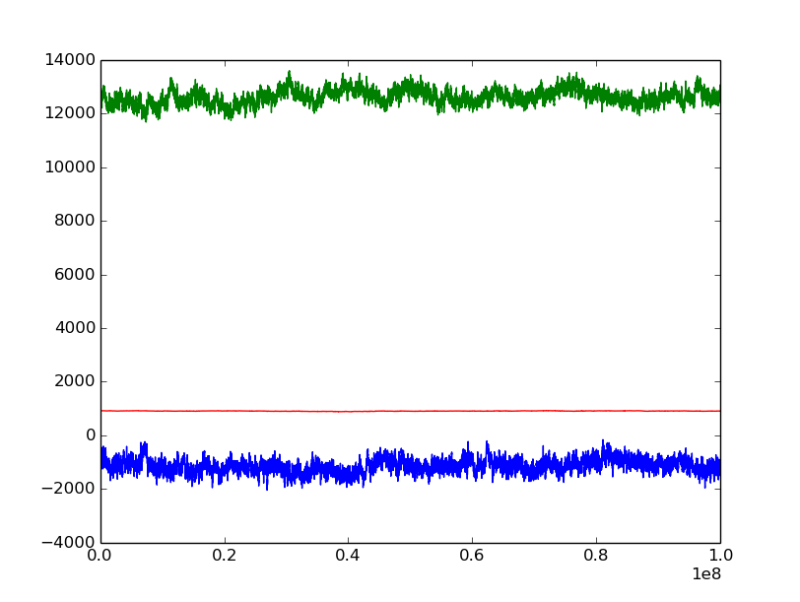
i analysed the 'U - mu * N' tendency with MC step, as illustrated in the figures
mu = -45.5 kJ/mol
mu = -47.5kJ/mol
the green line represents 'U - mu * N' [kJ], it seems that has reached to the equilibrium for both mu = -45.5 and mu = -47.5;
but the energy U and number of moleules N (not illustrated here) for mu = -47.5 are still decreasing, which also indicates that the water should be vapor at this mu
translation, rotation, insertion and deletion acceptance ratios are 0.25, would these be adjusted for a better simulation?
Kecheng
i analysed the 'U - mu * N' tendency with MC step, as illustrated in the figures
mu = -45.5 kJ/mol
mu = -47.5kJ/mol
the green line represents 'U - mu * N' [kJ], it seems that has reached to the equilibrium for both mu = -45.5 and mu = -47.5;
but the energy U and number of moleules N (not illustrated here) for mu = -47.5 are still decreasing, which also indicates that the water should be vapor at this mu
translation, rotation, insertion and deletion acceptance ratios are 0.25, would these be adjusted for a better simulation?
Kecheng
Please Log in to join the conversation.
- ryangmullen
-

- Offline
- Administrator
-

Less
More
- Posts: 124
- Karma: 4
- Thank you received: 24
5 years 6 months ago - 5 years 6 months ago #540
by ryangmullen
Hi Kecheng,
"it seems that has reached to the equilibrium" I wouldn't use any one thermodynamic property to gauge whether your simulation has converged. Rather, I'd want to see that all thermodynamic properties (e.g. U, N, and U-mu*N) have relaxed to fluctuate around a constant average value. Moreover, your final results should be independent of the initial configuration. So, if you have two simulations at the same muVT but different initial configurations that haven't converged to the same value, your only option is to run longer. But as we've discussed, near mu.equil for your temperature, those will be long simulations (billions if not trillions of MC moves).
So, what's your end goal? If you just want a liquid GCMC simulation at <P> = 1 bar, I think you have it with mu=-45.5 kJ/mol. If you want to map out the vapor-liquid equilibrium conditions, then the Gibbs ensemble would be a better choice than GCMC. At the very least, GEMC could tell you the <density> of the two phases at equilibrium.
Ryan
"it seems that has reached to the equilibrium" I wouldn't use any one thermodynamic property to gauge whether your simulation has converged. Rather, I'd want to see that all thermodynamic properties (e.g. U, N, and U-mu*N) have relaxed to fluctuate around a constant average value. Moreover, your final results should be independent of the initial configuration. So, if you have two simulations at the same muVT but different initial configurations that haven't converged to the same value, your only option is to run longer. But as we've discussed, near mu.equil for your temperature, those will be long simulations (billions if not trillions of MC moves).
So, what's your end goal? If you just want a liquid GCMC simulation at <P> = 1 bar, I think you have it with mu=-45.5 kJ/mol. If you want to map out the vapor-liquid equilibrium conditions, then the Gibbs ensemble would be a better choice than GCMC. At the very least, GEMC could tell you the <density> of the two phases at equilibrium.
Ryan
Last Edit: 5 years 6 months ago by ryangmullen.
Please Log in to join the conversation.
- FarawayK
-
 Topic Author
Topic Author
- Offline
- New Member
-

Less
More
- Posts: 6
- Thank you received: 1
5 years 6 months ago #541
by FarawayK
Hi Ryan,
thanks for your kindly reply and help, i've tried to run longer simulation based on different mu, some seems converge to my goal pressure, i would check them in the next days.
i'm planning to do simulation of water adsorption or condensation in nano pores, so before the formal simulation, i need do some pre trial to find the relation between mu and pressure or density. GEMC is a good way to find the pressure-density relation of liquid as you mentioned, but it's still unavailable to conduct GEMC simulation with fixed solid pore surface, so i choose GCMC to do this simulation. if there is better method to do simulation of water adsorption or condensation in nano pores, please tell me.
Kecheng
thanks for your kindly reply and help, i've tried to run longer simulation based on different mu, some seems converge to my goal pressure, i would check them in the next days.
i'm planning to do simulation of water adsorption or condensation in nano pores, so before the formal simulation, i need do some pre trial to find the relation between mu and pressure or density. GEMC is a good way to find the pressure-density relation of liquid as you mentioned, but it's still unavailable to conduct GEMC simulation with fixed solid pore surface, so i choose GCMC to do this simulation. if there is better method to do simulation of water adsorption or condensation in nano pores, please tell me.
Kecheng
Please Log in to join the conversation.
Time to create page: 0.199 seconds